 
MDR data alone cannot be used to establish rates of events, evaluate a change in event rates over time or compare event rates between devices.  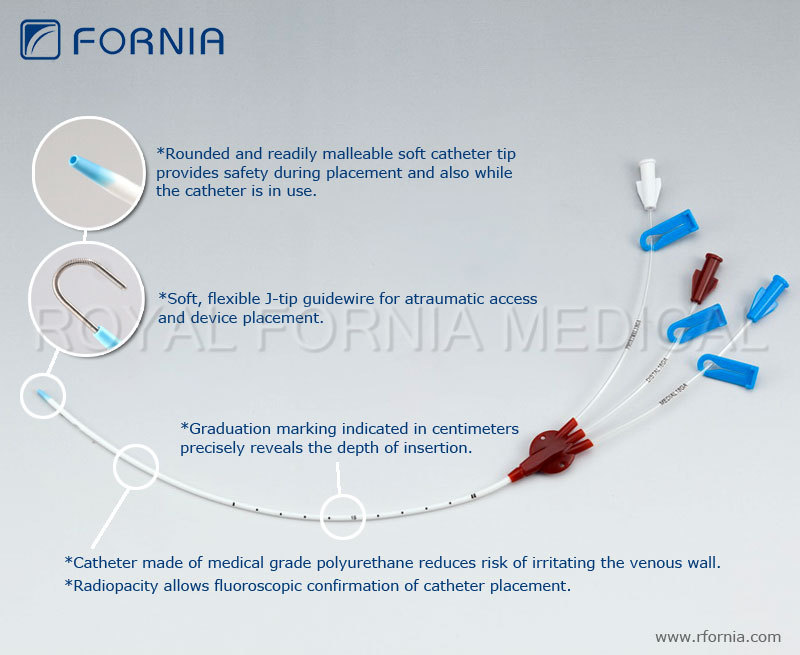 
Please note that the MAUDE web search feature is limited to adverse event reports within the past 10 years.Please see the MDR Data Files webpage for Device Experience Network (DEN) and Alternative Summary Report (ASR) data received by the FDA. Because of this, MDRs comprise only one of the FDA's several important postmarket surveillance data sources. In addition, the incidence or prevalence of an event cannot be determined from this reporting system alone due to under-reporting of events, inaccuracies in reports, lack of verification that the device caused the reported event, and lack of information about frequency of device use. The MAUDE database houses MDRs submitted to the FDA by mandatory reporters 1 (manufacturers, importers and device user facilities) and voluntary reporters such as health care professionals, patients and consumers.Īlthough MDRs are a valuable source of information, this passive surveillance system has limitations, including the potential submission of incomplete, inaccurate, untimely, unverified, or biased data. The FDA uses MDRs to monitor device performance, detect potential device-related safety issues, and contribute to benefit-risk assessments of these products. Arrow Select Kits Arrow® Central Venous Catheter Kit Arrow® Equipo de Cateterizacion Venosa Doble Lumen con Punta Flexible Azul Two-Lumen Central Venous Catheterization Set Arrow® HANDS-OFF® Multi-Lumen Central Venous Catheter with Blue FlexTip® Arrow® HANDS-OFF® Two-Lumen Central Venous Catheter with Blue FlexTip® Arrow® Marked Spring-Wire Guide with Arrow Advancer¿ Arrow® Multi-Lumen Central Venous Catheter with Blue FlexTip® Arrow® Multi-Lumen Central Venous Catheter with Blue FlexTip® for use only with Arrow MAC¿ Two-Lumen Central Venous Acce Arrow® Multi-Lumen Central Venous Catheter with Blue FlexTip® for use only with Arrow Percutaneous Sheath Introducer Arrow® Multi-Lumen Central Venous Catheterization Kit with Blue FlexTip® Catheter Arrow® Multi-Lumen Central Venous Catheterization Kit with Blue FlexTip® Catheter For Femoral Vein Insertion Arrow® Multi-Lumen Central Venous Catheterization Kit with Blue FlexTip® Catheter] Arrow® Multi-Lumen Central Venous Catheterization Kit with Blue FlexTip® Heparin-Coated Catheter Arrow® Multi-Lumen Central Venous Catheterization Set with Blue FlexTip® Catheter Arrow® Multi-Lumen Central Venous Catheterization Set with Blue FlexTip® Heparin-Coated Catheter Arrow® Multi-Lumen Central Venous Catheterization with Blue FlexTip® Catheter Arrow® Pediatric Multi-Lumen Central Venous Catheterization Set with Blue FlexTip® Catheter Arrow® Pediatric Two-Lumen Central Venous Catheterization Set with Blue FlexTip® Catheter Arrow® Percutaneous Sheath Introducer/Two-Lumen Central Venous Catheterization Kit Arrow® Quad-Lumen Central Venous Catheterization Set with Blue FlexTip® Catheter Arrow® Spring-Wire Guide with Arrow Advancer¿ Arrow® Two-Lumen Central Venous Catheter with Blue FlexTip® Arrow® Two-Lumen Central Venous Catheterization Kit with Blue FlexTip® Catheter Arrow® Two-Lumen Central Venous Catheterization Set with Blue FlexTip® ARROWg+ard Blue® Catheter Arrow® Two-Lumen Central Venous Catheterization Set with Blue FlexTip® Catheter CVC KIT: 2-LUMEN 7FR X 20CM CVC SET: 4-LUMEN 8.Each year, the FDA receives several hundred thousand medical device reports (MDRs) of suspected device-associated deaths, serious injuries and malfunctions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
